News
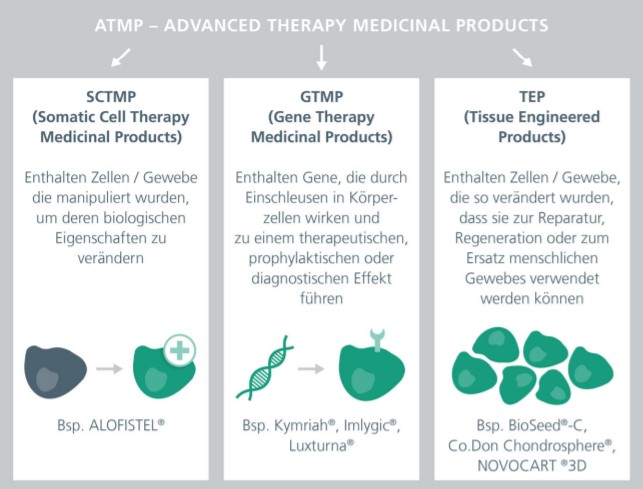
Advanced therapy medicinal products (ATMPs) are a new class of drugs comprising somatic cell therapeutics, gene therapeutics and bioengineered tissue products. To date, only a few such products have been approved throughout Europe. One reason for this is the lengthy and difficult path of their development. Prior to its use in humans, the safety of an ATMP must be demonstrated in a nonclinical phase in order to exclude potential side effects. Since ATMPs are of biological origin and thus exhibit high complexity and heterogeneity, individual testing strategies are required. The development of ATMPs from laboratory scale to manufacturing under Good Manufacturing Practice (GMP) conditions follows regulatory requirements.
To enable treatment of larger patient populations with ATMPs, GMP processes need to be scaled up, which is a major logistical and technological challenge. The development of ATMPs is costly and time-consuming but also offers unique new opportunities for the treatment of severe diseases, such as cancer.
Regulatory approval of an ATMP normally first requires non-clinical studies to be conducted in the regulated environment of a GLP testing facility. In Germany, besides various commercial organizations, five institutes of the Fraunhofer-Gesellschaft (Fraunhofer Institute for Biomedical Engineering IBMT, Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB, Fraunhofer Institute for Translational Medicine and Pharmacology ITMP, Fraunhofer Institute for Toxicology and Experimental Medicine ITEM, and Fraunhofer Institute for Cell Therapy and Immunology IZI) are certified GLP testing facilities.
New publication “ATMP: the road to clinical application”:
Neuhaus, V., Ueberham, E., Dehmel, S. et al. ATMP: the road to clinical application. Forum (2021).
Read the paper (German language, abstract in English) https://doi.org/10.1007/s12312-021-00995-0
 Fraunhofer Institute for Toxicology and Experimental Medicine
Fraunhofer Institute for Toxicology and Experimental Medicine