Practicability of phage therapy in Germany
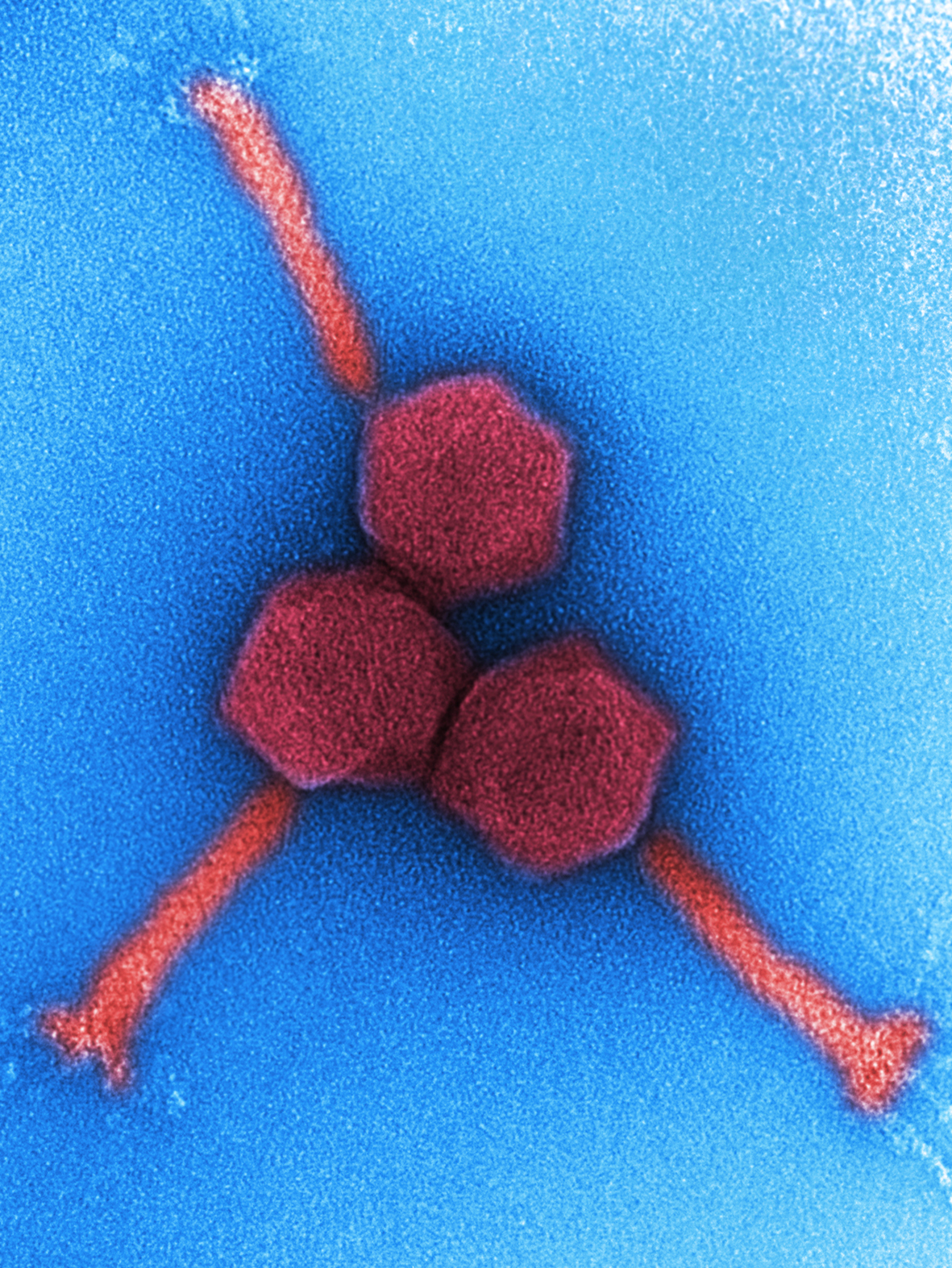
The aim of the project PhagoFlow in Braunschweig is to determine the infrastructural requirements for magistral production of phages in Germany to enable treatment of patients with wound infections of the extremities. This project, initiated by the Federal Joint Committee and funded by the Innovation Committee of the German Bundestag, is a collaboration of Fraunhofer ITEM, Leibniz Institute DSMZ and the Bundeswehr Hospital in Berlin.
As a first step, DSMZ identified 12 phages targeting different WHO prioritized pathogens based on their biological properties: complementary host range, high lytic activity and absence of undesired genes. These phages then were to be produced for magistral use. The challenge in this context, however, is the lack of a regulatory guideline and drug monograph. The ITEM researchers, therefore, focused at first on the definition of minimum requirements for quality control testing, GMP aspects and the purity of phage preparations and on discussing these with the competent authorities. The aim was to combine a high level of patient safety with cost-effective and time-saving production.
This was followed by pilot production of the most promising phages based on the previously established GMP and pharmaceutical quality requirements. Individualized phage preparations shall then be formulated by the Bundeswehr Hospital Pharmacy in Berlin based on phagograms (phage susceptibility tests against pathogens) and are planned to be administered to selected patients in the Department of Trauma Surgery.
 Fraunhofer Institute for Toxicology and Experimental Medicine
Fraunhofer Institute for Toxicology and Experimental Medicine